Research
|
The Roh Lab studies mechanisms that control cellular identity and plasticity, seeking to use our findings to cure disease and improve human health.
|
Adipose biologyAdipocytes play a key role in nutrient and energy homeostasis, directly implicated in metabolic disease, such as obesity and diabetes. We have previously made an unexpected finding that, despite being a terminally differentiated post-mitotic cell type, adipocyte has potentials to reprogram its cellular identity in response to physiological and environmental cues.
Adipose biology is our major research project. We are currently working on two projects. (1) We study how adipocyte control its color (white, brown, and beige) in response to temperature changes. We are especially interested in transcriptional and epigenetic mechanisms that drive temperature-responsive adipocyte reprogramming. We seek to use our findings to enhance energy metabolism and to improve metabolic health. (2) We recently discovered that adipocytes adopt altered cellular states with the emergence of muscle-like signatures during obesity. We work to understand how this change is regulated and how it contributes to adipose tissue function and systemic energy metabolism. |
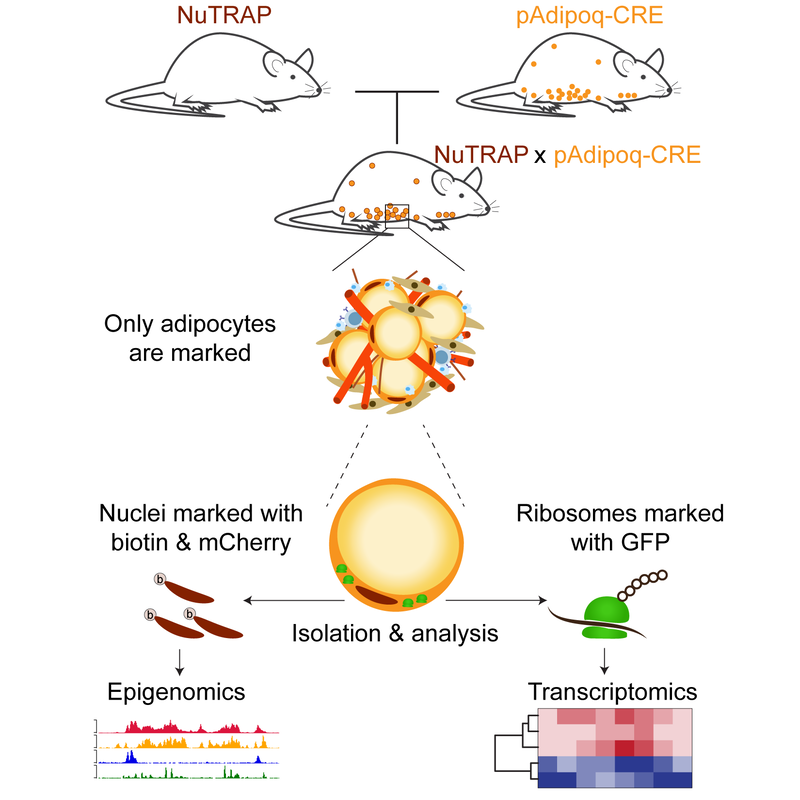
EpigenomicsWe have previously developed a new transgenic mouse, called NuTRAP, which allows for cell type-specific transcriptomic and epigenomic analysis in vivo. NuTRAP mice has led us to the discovery of adipocyte plasticity.
We continue to employ our NuTRAP mice to elucidate epigenetic remodeling underlying adipocyte plasticity. In addition to RNA-seq and ChIP-seq, we are working with NuTRAP for cutting-edge epigenomic profiling techniques, such as ATAC-seq, and CUT&Tag. We hope to extend to the methods for higher-order chromatin conformation and nuclear architecture analysis. We also work on single cell & nucleus sequencing analysis. We aim to identify cellular heterogeneity in adipose tissues and to address cellular plasticity and population dynamics in response to physiological and environmental cues. |
Cellular plasticityOnce differentiated, somatic cells maintain their cellular identity and fulfill unique cellular functions. Our studies with adipocytes, however, suggest that other somatic cell types may also have potentials to switch cellular identity. We are exploring whether and how such cellular plasticity contributes to normal physiology of other tissues and is implicated in human disease.
We are collaborating with other investigators on the campus to test this possibility. |